Five years have passed since we first shared with you that twin boys affected by X-linked hypohidrotic ectodermal dysplasia (XLHED) were sweating after receiving an investigational treatment before birth. It’s hard to believe how quickly time has flown!

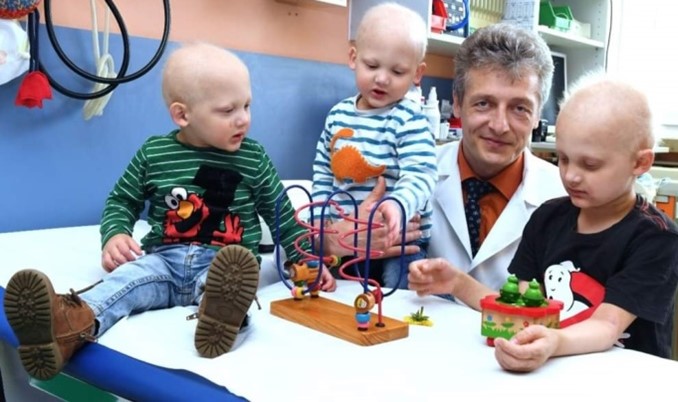
Do you remember Maarten and Linus? Today, I’m excited to share with you an update on how the 7-year-old twins (see photo at top of page) and the other boys who were treated are doing.
Prof. Dr. Holm Schneider at the University Hospital Erlangen leads this study. Earlier this year, he and his team published their latest findings in the International Journal of Molecular Sciences. I will share with you key highlights from their long-term, follow-up study.
Background
XLHED is caused by the lack of a protein called ectodysplasin A1 (EDA1). This protein is what is essential for the development of sweat glands and hair follicles, teeth, mucous membranes in the upper respiratory tract, salivary glands, and lacrimal and meibomian glands in the eye.
Researchers created a replacement protein that mimics EDA1. From 2016 through 2021, Dr. Schneider treated six babies in utero, who were affected by XLHED, with this recombinant protein which is called ER-004. He injected it into the amniotic fluid of the pregnant women. It was swallowed by the fetuses, during the natural time window when their sweat glands would develop.
All but one of the babies received at least two doses of the protein in utero, with the other one receiving one dose only. The study also includes data from three boys affected by XLHED who received ER-004 after birth for comparison.
Long-Term Results
The study followed the boys until they were five years old and tracked key data. Dr. Schneider’s team reported the following findings:
Sweat Glands
All of the boys who received at least two doses had a normal number of sweat pores! These five also all had a normal or near-normal amount of sweating when chemically stimulated.
These results matched what their parents reported: the boys were repeatedly sweating on different parts of their body. Efficient thermal sweating became nicely evident in the treated twins who were studied when having a sauna with their father.
According to the publication, the oldest two boys have perspired normally every year. They have been able to do normal outdoor activities with high levels of fitness. None of the boys have experienced overheating (hyperthermia).
Eye/Ear/Nose/Throat/Respiratory
None of the boys have had any significant eye, nose, throat or respiratory issues nor did they have hospitalizations. This is extraordinary news as respiratory issues typically can be very challenging in little ones affected by XLHED.
Teeth
Findings show that the protein treatment does not impact the primary (baby) teeth. The protein does appear to impact the development of permanent teeth but it’s not optimal. The treated boys have more permanent teeth than their brothers who have XLHED and are untreated. But, they do not have a full set of teeth. The research team will need to study their permanent dentition for a longer time before they can fully know how the treatment influenced their teeth.
Hair
The recombinant protein has not improved the boys’ scalp hair development. We expected that result since hair follicle formation takes place very early in a pregnancy.
What We Learned
Overall, the news is very positive.
- The long-term data reinforced earlier findings that administering the protein after birth did not improve XLHED symptoms.
- Administering ER-004 at the right time before birth can lead to normal sweat gland development.
- Giving the treatment via intra-amniotic injection is effective and safe.
- The treated boys can regulate their core body temperature normally which will positively affect their ability to go to school and participate in physical activities.
- The treated boys have not had any hospitalizations for XLHED symptoms.
Some women have asked us if the treatment effect can wear off over time. Dr. Schneider wrote in the journal article:
To our knowledge, there is no reason to assume that once sweat glands are normally formed, they would not work permanently.
– Prof. Dr. Holm Schneider
EDELIFE Clinical Trial – How You Can Participate
We are thrilled that the six boys are all experiencing long-term improvement of symptoms! Our hope is that this treatment will one day be on the market as an option for all XLHED families.
But first, the EDELIFE clinical trial must confirm these initial, promising findings. If your family is affected by XLHED, you or your loved one may be eligible to participate in this important clinical trial which is now available in six countries. The trial is enrolling patients and five families are currently participating.
The timing of administering the investigational medicine during the pregnancy is critical. If you are pregnant or planning to be, we recommend that you learn more about the trial now.
You can learn more on our site and the EDELIFE clinical trial site. Also, feel free to contact me at any time. I would be happy to share more details with you about EDELIFE.
Learn MoreTogether, We Can Do This!
I extend my gratitude to all of the XLHED families who have participated in research studies for 30+ years that led to this trial. Even if you are not eligible for the trial, you can help by sharing information about it with your family and on your socials.

Congrats to the researchers!
This is incredible news, especially for the boys and their parents!!